3DEXPERIENCE ENOVIA Role
Regulatory Team Member (GLR)
Digitizes and institutionalizes best practices for managing product market authorization and post market surveillance
Get Started on 3DEXPERIENCE.
- Configured Best Practice
Digitize and institutionalize best practices for registering new products with pre-configured best practice templates to support health authorities - Product Registration Lifecycle Management
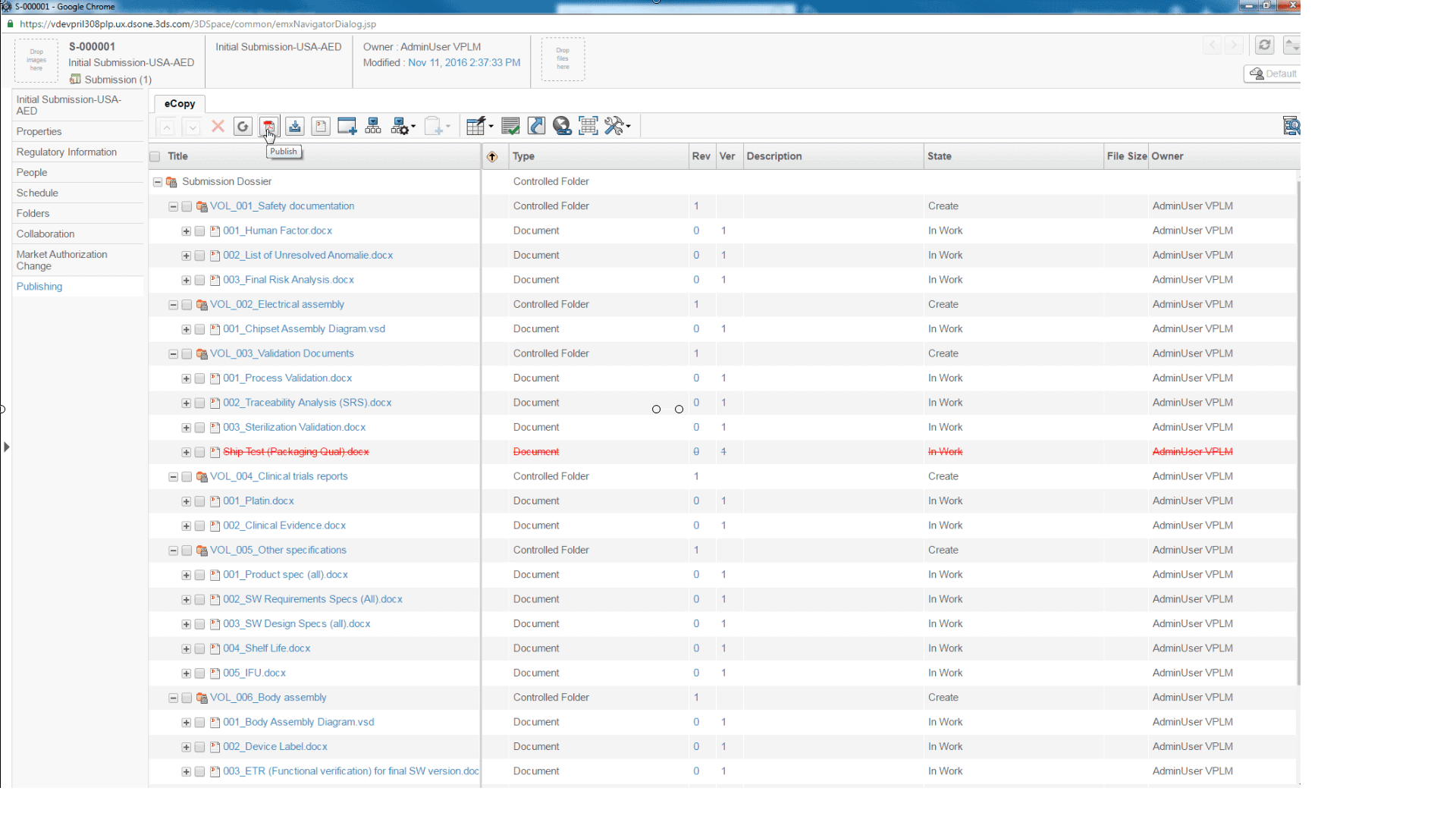
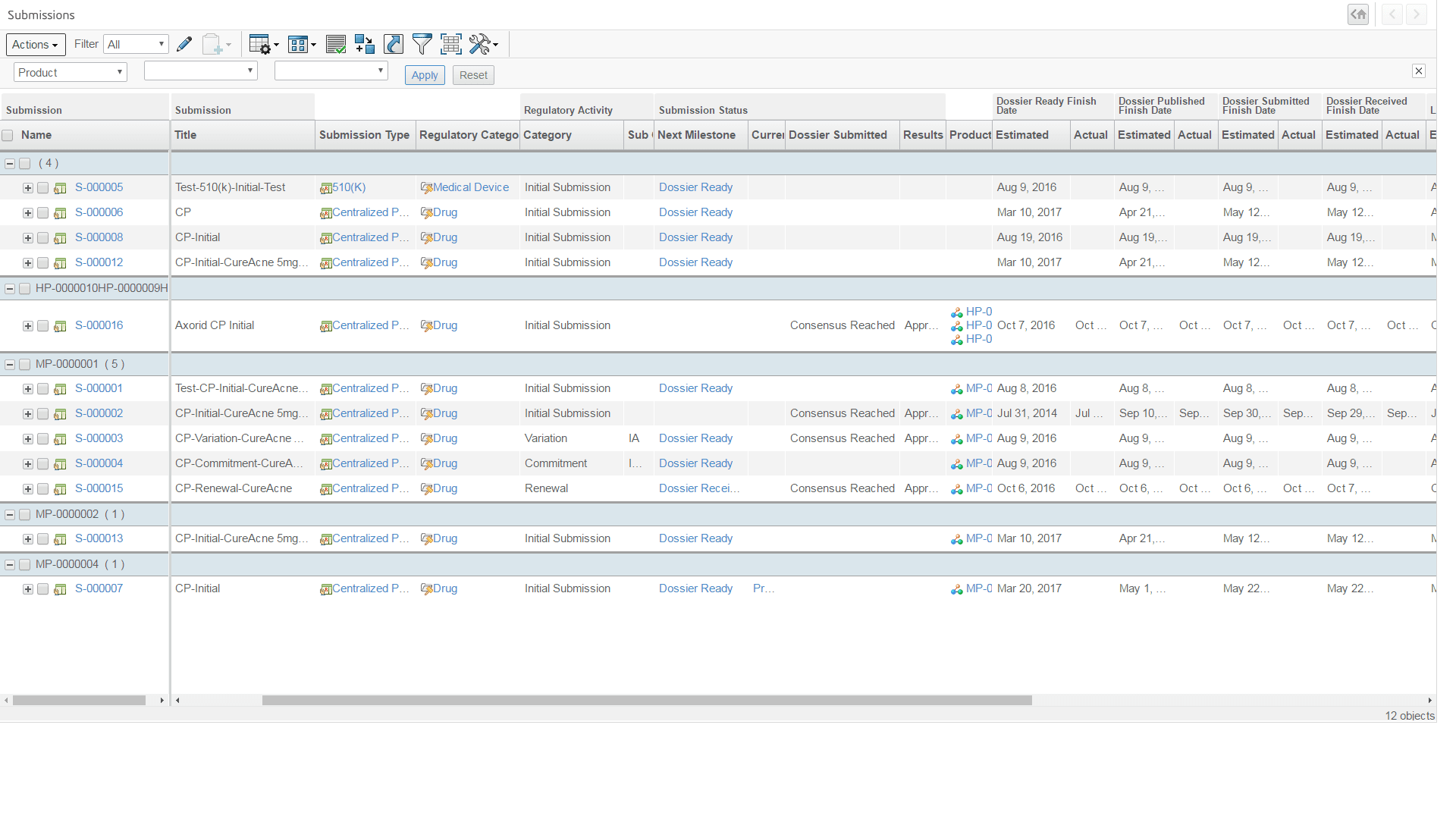
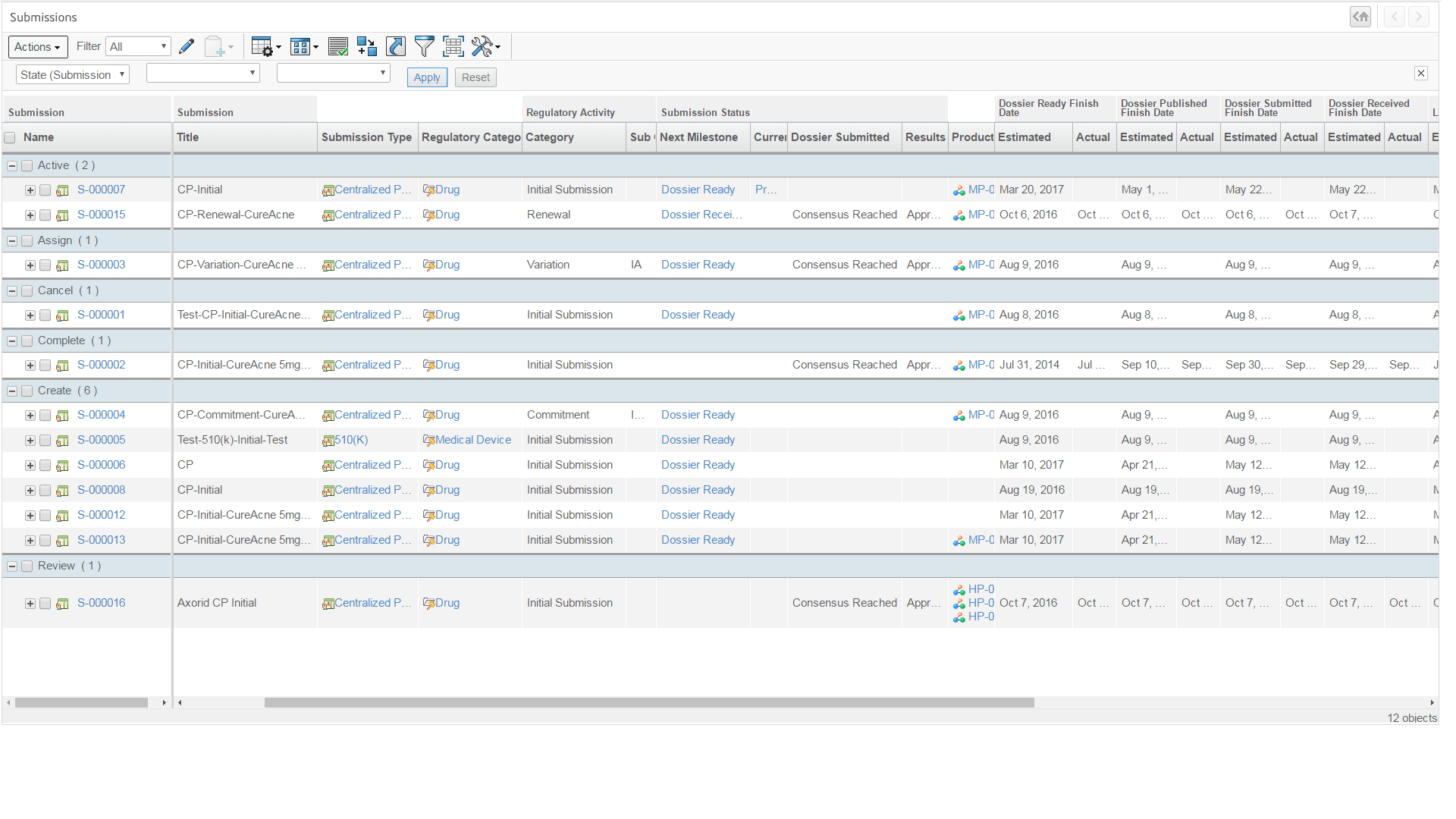
Manage product registrations lifecycle & approvals from planning, document collection, submission assembly, interactive review to achieve market authorization - Product Safety Reporting
Evaluate and manage product safety issue to determine reportability with support for electronic and paper based safety report to authorities - Medical Device Unique Identifiers (if used for Medical Device)
Manage the Medical Device identification record and registration processes electronically to the FDA GUDID
4 Included Apps
Adverse Event Reporting
Device Identification
Market Registration
My Submissions
3DEXPERIENCE Learning and Support
3DEXPERIENCE Learning Resources
3DEXPERIENCE is powerful and transformative software, so learning to make the most of it isn’t trivial. Thankfully, Dassault Systèmes and CATI provide plenty of resources on the path to proficiency:
- Official and custom-tailored classroom training
- Online training courses on the Learning Space
- CATI’s quick-response technical support
- Extensive official documentation
- Dassault Systèmes Knowledge Base of supplemental materials, technical articles, and support tickets
- CATI simulation mentorship programs
- CATI consultative methodology development
Why 3DEXPERIENCE with CATI?
Computer Aided Technology (CATI) has been implementing solutions on the 3DEXPERIENCE Platform since 2012. No one has more experience when it comes to 3DEXPERIENCE Platform solutions, especially when it comes to implementing the platform with SOLIDWORKS or CATIA.
Watch this video to learn more about why companies choose to partner with CATI for 3DEXPERIENCE solutions and get started today.
Get Started with 3DEXPERIENCE from CATI.
We’re ready to help figure out the right product development solution plan for you. Contact us today to get started with a quote, a trial, a demo, or just ask questions.