SOLIDWORKS Simulation: Virtual Testing for OxyArm
OxyArm: Engineering a Medical First with Simulation
How many problems do you need to solve while bringing a new product to market? Probably more than you want to think about right now. However, despite setbacks, looming deadlines and unforeseen circumstances, you manage to get the job done. When it’s over, you feel great. Well, I want to make it possible for you to feel better earlier in the design process with help from virtual testing.
In this multi-part blog series, I’m going to take you through the testing process used by Southmedic, Inc. on its no-contact oxygen delivery device called the OxyArm, which was the first open oxygen delivery system on the market. To make this new system a reality, Southmedic used CAD embedded virtual testing while designing the OxyArm. This series will cover how SOLIDWORKS Simulation features were used to determine optimal air flow, best materials for plastic injection molding, calculate the device’s durability and understand cost to manufacture.
This post will focus on the most important piece of the puzzle: making sure the product will work as intended. During the conceptual phase, Southmedic found that traditional oxygen masks made users feel claustrophobic, created unpleasant odors and caused nasal burns, chafing, and sinus sensitivity. These findings pushed the company to develop a product that would deliver oxygen through a headset and eliminate these areas of dissatisfaction. In the case of an oxygen mask, ensuring that it delivers the optimal amount of air is paramount.
Testing how air would flow through the OxyArm required setting up two design studies with SOLIDWORKS Flow Simulation: a diffusor optimization and a breathing simulation to look at the amount of oxygen that will be delivered to the user’s mouth. First, let’s investigate the diffusor optimization, which determined the best length for the stem used to distribute air evenly to a user’s nose and mouth.
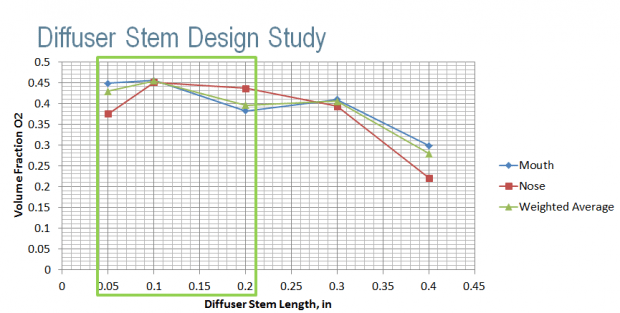
The first step of the investigate just the length of the stem, varying it from 0.05” – 0.40”, the results below show the air velocity and the volume fraction of oxygen delivered to the mouth of a user. The chart shows that the optimum length is in the 0.05” – 0.2” region.
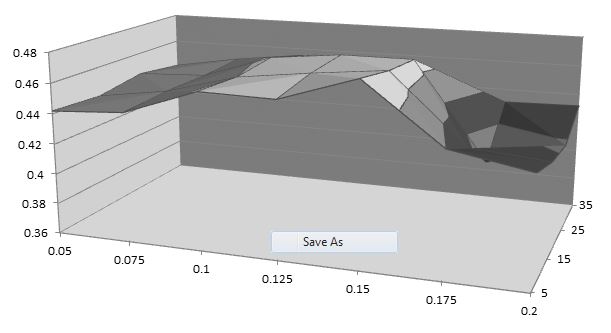
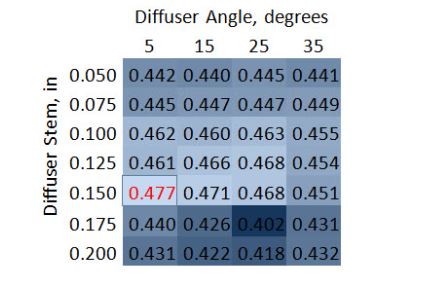
But, how often do we just need to optimize for one variable, like length in this case. Flow Simulation doesn’t constrain the number of variables we can use to run a design study, so you can take this further and look at a two variable optimization with length and tip angle. The data below shows that the optimum configuration is a diffuser length of 0.15” and angle of 5o.
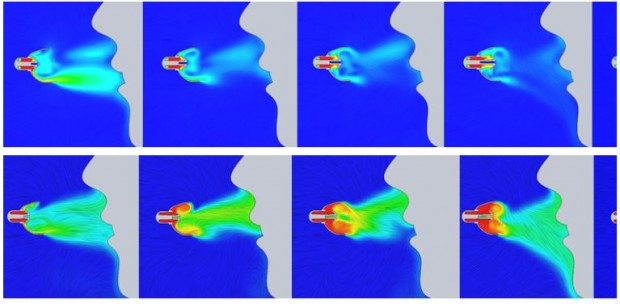
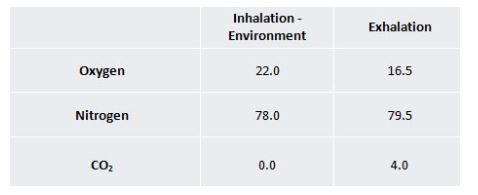
Now the mask has been optimized for steady state flow performance, you can take a step further and use virtual testing to understand how the OxyArm will behave during a multiple breathing cycles. This allows for the visualization of exhalation impact on oxygen concentrations around the mask. The breathing specs are shown below, environmental conditions are shown in the inhalation and the exhalation has well known concentration values.
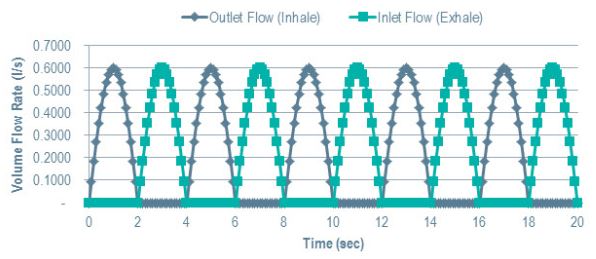
You can also specify transient inhale/exhale properties, and fully investigate the transient response.
For the OxyArm, flow simulation enabled engineers to determine both the best size diffusor for optimal airflow performance and how the device would react while in use to gain an understanding of its operational environment. Next up in process for creating the first open oxygen delivery system is testing the best materials for injection molding. Stay tuned for the next post to see how Southmedic used virtual testing to investigate different plastic materials and select the best option for the OxyArm.
To learn more about medical device design and overcoming regulatory and time constraints, check out our life sciences page to read white papers, see ProNova’s cancer-killing cyclotron in action, hear from customers and learn how you can bring your next medical breakthrough to market in record time.

 Blog
Blog